Tracking and balancing your pool water—and what could happen if you don’t!
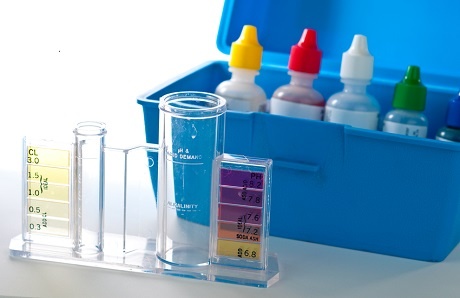
Regular pool water testing and maintenance of the chemicals are essential for ensuring a safe, comfortable swimming environment and preserving the longevity of pool finishes and equipment.
Failure to monitor and maintain water quality can lead to damage or deterioration to the pool features, materials, and equipment. Above all, it can cause water conditions that may harm swimmers and cause a variety of illnesses.
Avoid the hassles, hazards, and risks to those using your pool by keeping water properly maintained. Testing the chemistry is simple. Make it part of your routine, so you have the needed data to treat water for a healthy aquatic retreat.
Methods available for testing pool water
The chief products for analyzing pool water chemistry are test strips, 4-in-1 test kit, and testing tablets. Each one can provide a dependable reading for chemical levels in pool water, and most include step-by-step instructions that are simple to follow for first-time pool owners.
So, which compounds do you need to test for? Let’s take a look at the four key chemicals essential for a trouble-free, swim-ready environment.
Pool-water chemical levels to test and balance
The four chemical levels critical to healthy water are free chlorine, calcium hardness, pH, and total alkalinity. Along with cyanuric acid (a type of water conditioner sometimes referred to as “sunblock for pool water”), these compounds work in unison, and having each at the right level helps deliver sparkling, swim-ready water.
If water analysis indicates that one or more chemical levels are too high or too low, the message is to take action. With the water readings, you will know to add products like chlorine, muriatic acid, sodium bicarbonate, or other pool treatments.
Free Chlorine
Whether tablet, liquid, or powder form, the chlorine in a pool kills an array of viruses, bacteria, and organic matter such as sweat, urine, and fecal matter. Chlorine disinfects the water to remove most pathogens that can harm swimmers.
Free chlorine levels refer to the chlorine in pool water that is “fresh”—it has yet to react with any contaminants. Too much or too little of this chemical in the water can be potentially harmful to swimmers and your pool equipment.
Without enough free chlorine to attack invading contaminants, pool water becomes susceptible to a variety of undesirable effects.
Algae may grow, turning the water murky with slimy green patches along the pool’s floor and walls. The unwanted green visitor can also coat the cartridges in some kinds of pool filters—necessitating more frequent cleaning that shortens product lifespan.
Without sufficient chlorine, the water’s appearance may also become cloudy from organic matter that the wind blows in and swimmers bring in, such as sweat, make up, body oils, and suntan oil.
Worse yet, too little chlorine gives safe harbor to water-borne diseases! When swimmers (including pets) introduce biological matter—from urine to bacteria infecting an open sore—there is not enough sanitizer to defeat the pathogens. As a result, opportunities increase for transmission of infectious microbes and the illnesses they cause.
On the flip side, high levels of free chlorine cause its share of issues. Too much can sting swimmers’ eyes and irritate their skin. Elevated levels of this chemical can cause harsh, bleach-like fumes that are downright nasty to inhale and irritating to swimmers’ lungs.
Pools need enough free chlorine to kill unwanted pathogens and contaminants, but not so much that is poses noxious effects to pool users. It’s all about achieving balance with a range of 1.5 to 2.5 ppm. Note: Pools with ozone systems and/or ultraviolet sanitizers require dramatically less chlorine—only .5 to 1 ppm.
pH
The pH level represents the total acidity of pool water. It’s measured on a scale of 1 to 14: 1 through 6 indicate how much acidity; 8 through 14 indicate alkalinity. Right in the middle—7.3 to 7.7 ppm—is neutral, and where pool water should be.
If the pH level is too low, water can irritate swimmers’ eyes and skin. Low pH can damage pool equipment as well: Ladders, railings, and other accessories with metal parts can begin corroding, as can parts of the pool heater.
If the pH is too high, it can lower the effectiveness of the free chlorine in water—which may result in unsanitary swimming conditions. A high pH range may cause water to become cloudy, and like water with low pH, it may make skin itch and eyes burn.
If the alkalinity and calcium hardness levels in the pool are elevated as well, the resulting combination with high pH means other troubles. The imbalanced chemical triad can cause rough scale on the pool’s skimmer, interior surfaces, various equipment fittings, and on part of some kinds of automatic pool cleaners.
Total alkalinity
Total alkalinity (TA) refers to the total amount of alkaline substances present. For TA to be effective in a pool the level should fall between 90 and 120 ppm.
Alkaline is important to pool-water chemistry as it has a major impact on keeping the pH level neutral. If the TA level is too high or too low, pH levels may fluctuate; often, if the TA level is off, the pH will be off as well.
Low TA is problematic. Ramifications to anyone using the pool include burning eyes and having to swim in dark murky water.
The pool bears the major impact. The water becomes “aggressive,” and attacks metal parts of pool accessories and equipment like railings, ladders, and the heat exchanger in the heating system.
On the other hand, a high TA level in the water can irritate swimmers’ skin. It can also reduce free chlorine's effectiveness—which can spur algae and bacteria growth, and the entire host of issues that come with low chlorine levels.
Calcium hardness
Calcium hardness measures the hardness level of pool water, indicating if water is too hard, too soft, or just right.
If the hardness level is too low—meaning the water is soft—pool plaster bears the brunt. Desperate for calcium, the pool water absorbs needed calcium from the best-available source—the pool plaster! As the mineral leeches out of the materials, the previously smooth interior finish becomes etched and full of unsightly pitting.
Of course, if the calcium hardness is too high, it ushers in another set of problems. Ugly scaling—deposits of calcium—can build up on pool’s interior surface, producing gray, white, or brownish spots throughout the pool’s interior.
If the pH and alkalinity are also elevated like the calcium, the news is particularly bad. Scale can build up and wreak havoc on components in the pool pump, heater, and DE or cartridge filter.
Extreme cases of scaling that have gone unchecked may require a costly, cumbersome endeavor any pool owner wants to avoid: draining the pool. That’s what’s required to perform and acid wash it to remove all of the mineral build up.
Don’t let that happen. To prevent out-of-control calcium and associated complications, check that the calcium levels range from 200 – 400 ppm.
The Takeaway
Testing your pool water and keeping chemicals in balance is paramount for the lifespan of your pool and the health and safety of your friends and family members who swim in it.
Toward this goal, regularly test your water on a set weekly schedule to ensure levels remain balanced. Properly maintaining pool chemistry will help you and your family enjoy your backyard resort for years to come!

.jpg?width=1490&name=rock-waterfall-slide-pool%20(1).jpg)